Turbinoliidae
Stephen D. Cairns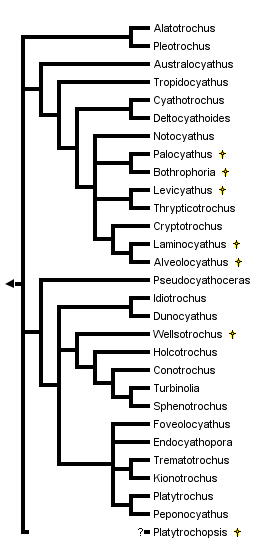

This tree diagram shows the relationships between several groups of organisms.
The root of the current tree connects the organisms featured in this tree to their containing group and the rest of the Tree of Life. The basal branching point in the tree represents the ancestor of the other groups in the tree. This ancestor diversified over time into several descendent subgroups, which are represented as internal nodes and terminal taxa to the right.

You can click on the root to travel down the Tree of Life all the way to the root of all Life, and you can click on the names of descendent subgroups to travel up the Tree of Life all the way to individual species.
For more information on ToL tree formatting, please see Interpreting the Tree or Classification. To learn more about phylogenetic trees, please visit our Phylogenetic Biology pages.
close boxIntroduction
The turbinoliids are known from the Late Cretaceous (Campanian of Antarctica; Haumurian of New Zealand) to the Recent, spanning 75 million years. Living representatives are widespread in the world oceans, but are not known from off continental Antarctica, most of the eastern Pacific, and cold temperate northwest Atlantic. They are particularly diverse and abundant in the Indo-West Pacific region, especially off southern Australia, New Zealand, and the Indonesian regions. They live at depths of 6 to 1137 m. Turbinoliids are exclusively azooxanthellate and solitary in growth form, rarely more than 10 mm in size. Indeed, Turbinolia stephensoni, which was originally described in the genus Oryzotrochus by Wells (1959) because of its resemblance in size and shape to a grain of rice (Greek oryza = rice), measures only 1.5 mm in calicular diameter. The turbinoliids have one of the highest generic diversities among the Scleractinia (28, including 22 Recent), and the family contains 163 valid species (Cairns, 1997), only 51 of which are Recent (Cairns et al., 1999).
Because turbinoliid coralla are so small and infrequently collected, little is known about their biology. It is known that all turbinoliids completely cover, or invest, their corallum with tissue, making permanent attachment to a substrate impossible. The complete investiture of the turbinoliid corallum may facilitate movement through or across a sandy medium, as is the case of certain mushroom corals (Chadwick, 1988; Hoeksema, 1993) and thus, might be interpreted as an adaptation to an interstitial or semi-burrowing habit in sandy substrates at lower shelf and upper slope depths -- a niche exploited by few other Scleractinia most of which require a hard substrate for original planular settlement and subsequent support. Turbinoliids live in sandy-shelly environments, incorporating and overgrowing small bits of sand and shell into their base.
The paleoecology of turbinoliids was discussed by Filkorn (1994: 13-16) and the family was revised, including illustrations of all type species, by Cairns (1997). At least one genus, Peponocyathus, is the host for the galls of parasitic ascothoracidan Crustacea (Grygier and Zibrowius, 1985).
Characteristics
Turbinoliids have septothecal walls that are never reinforced with epitheca or textura. Most have well-developed costae and deep intercostae furrows, a result of the complete investiture of the corallum by the polyp. The intercostal regions may be solid, pitted, or perforate. The genera have a great diversity of shapes, palar configurations, and columellar types, which form the basis of generic differentiation. Also, four genera reproduce by transverse division; four genera bear twice as many costae as septa; and, in two genera, the costae are offset from the septa.


Dunocyathus parasiticus: The anthocyathus (distal portion) of a skeleton collected off Cape Jaffa, South Australia (depth 165 m). Note that in this species the costae alternate in position with the septa. Calicular diameter 4.4 mm. From Cairns & Parker, 1992, copyright © 1992 South Australian Museum.
Discussion of Phylogenetic Relationships
A phylogenetic analysis of the turbinoliid genera was done recently (Cairns, 1997), resulting in 5876 equally parsimonious trees. Successive weighting of characters and other logical assumptions resulted in the one tree presented above. This is not the consensus tree, but one of the 5876 equally parsimonious trees, and it is not considered to be very robust. Nonetheless, this phylogenetic hypothesis suggests that the turbinoliids are divided into two major clades. One clade (Australocyathus to Alveolocyathus) contains 12 genera including all six late Cretaceous Antarctic genera, as well as genera first recorded from the Eocene of New Zealand and Oligocene of South America. Genera of this clade are characterized by having trifurcate costal origination and serrate costal ornamentation. The other major clade (Pseudocyathoceras to Peponocyathus) contains 14 genera, including one from the Late Cretaceous of New Zealand, five with first occurrences in the Paleocene to Miocene of Europe and North America, and three from the Eocene to Oligocene of South America. These are characterized by coralla with fewer than 48 septa and granular or smooth costae.
References
Cairns, S. D. 1979. The deep-water Scleractinia of the caribbean Sea and adjacent waters. Studies on the fauna of Curaçao and other Caribbean Islands, 57: 341 pp.
Cairns, S. D. 1994. Scleractinia of the temperate North Pacific. Smithsonian Contributions to Zoology, 557: 150 pp.
Cairns, S. D. 1997. A generic revision and phylogenetic analysis of the Turbinoliidae (Cnidaria: Scleractinia). Smithsonian Contributions to Zoology, 591: 55 pp.
Cairns, S. D., B. W. Hoeksema, and J. van der Land. 1999. Appendix: List of Extant Stony Corals. Atoll Research Bulletin, 459: 13-46.
Cairns, S. D. and S. A. Parker. 1992. Review of the Recent Scleractinia (Stony Corals) of South Australia, Victoria and Tasmania. Records of the South Australian Museum, Monograph Series, 3: 82 pp.
Chadwick, N. E. 1988. Competition and locomotion in a free-living fungiid coral. Journal of Experimental Marine Biology and Ecology, 123: 189-200, 2 figs.
Filkorn, H. F. 1994. Fossil scleractinian corals from James Ross Basin, Antarctica. Antarctic Research Series, 65: 96 pp.
Grygier, M. J. and H. Zibrowius. 1985. Diversity and range of scleractinian coral hosts of Ascothorida (Crustacea: Maxillopoda). Annals de l'Institut Océanographique, Paris, 61(2): 115-138.
Hoeksema, B. W. 1993. Phenotypic corallum variability in Recent mobile reef corals. Courier Forschungsinstitut Senckenberg, 164: 263-272, 4 figs.
Wells, J. W. 1959. Notes on Indo-Pacific scleractinian corals, Part 1. Oryzotrochus, a new genus of turbinolian coral. Pacific Science, 13: 286-287.
Title Illustrations

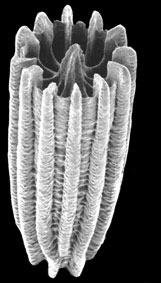
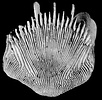
| Scientific Name | Turbinolia stephensoni |
|---|---|
| Location | Arafura Sea (depth 24 m) |
| Comments | This species has the smallest adult corallum of any scleractinian, this specimen being only 2 mm in height |
| Reference | Cairns, S. D. 1997. A generic revision and phylogenetic analysis of the Turbinoliidae (Cnidaria: Scleractinia). Smithsonian Contributions to Zoology, 591: 55 pp. |
| Specimen Condition | Dead Specimen |
| Body Part | skeleton |
| View | Oblique SEM view |
| Size | 2 mm high |
| Image Use |
 This media file is licensed under the Creative Commons Attribution-NonCommercial License - Version 3.0. This media file is licensed under the Creative Commons Attribution-NonCommercial License - Version 3.0.
|
| Copyright |
© 1997

|
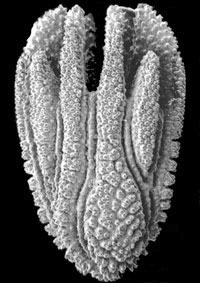
| Scientific Name | Holcotrochus scriptus |
|---|---|
| Location | Kangaroo Island, South Australia (depth 108 m) |
| Comments | This is one of the smallest of the scleractinian corals (height 2.3 mm) and has a unique shape and costal morphology. |
| Reference | Cairns, S. D. and S. A. Parker. 1992. Review of the Recent Scleractinia (Stony Corals) of South Australia, Victoria and Tasmania. Records of the South Australian Museum, Monograph Series, 3: 82 pp. |
| Specimen Condition | Dead Specimen |
| Body Part | corallum |
| Copyright | © 1992 South Australian Museum |
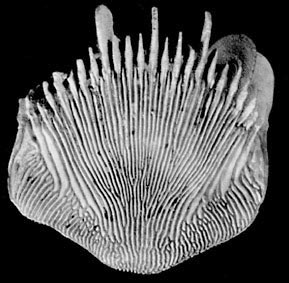
| Scientific Name | Alatotrochus rubescens |
|---|---|
| Location | Kyushu Island, Japan (depth 193 m) |
| Comments | characterized by having prominent edge crests and twice as many costae as septa |
| Reference | Cairns, S. D. 1994. Scleractinia of the temperate North Pacific. Smithsonian Contributions to Zoology, 557: 150 pp. |
| Body Part | skeleton |
| View | Side view |
| Size | Height of corallum 14 mm |
| Image Use |
 This media file is licensed under the Creative Commons Attribution-NonCommercial License - Version 3.0. This media file is licensed under the Creative Commons Attribution-NonCommercial License - Version 3.0.
|
| Copyright |
© 1994

|
About This Page
Creation of this page was supported by US National Science Foundation grants DEB95-21819 and DEB 99-78106 (in the program PEET - Partnerships to Enhance Expertise in Taxonomy) to Daphne G. Fautin, grant DEB99-78086 (in the program PEET) to Stephen D. Cairns, and grant OCE 00-03970 (in NOPP, the National Oceanographic Partnership Program) to D.G.F. and Robert W. Buddemeier.
Technical assistance was rendered by Adorian Ardelean.
The author welcomes the opportunity to identify specimens from this family, and offers to incorporate them into the collections of the National Museum of Natural History, Smithsonian, unless their return is requested.

Smithsonian Institution, Washington, D. C., USA
Correspondence regarding this page should be directed to Stephen D. Cairns at
Page copyright © 2002
 Page: Tree of Life
Turbinoliidae .
Authored by
Stephen D. Cairns.
The TEXT of this page is licensed under the
Creative Commons Attribution-NonCommercial License - Version 3.0. Note that images and other media
featured on this page are each governed by their own license, and they may or may not be available
for reuse. Click on an image or a media link to access the media data window, which provides the
relevant licensing information. For the general terms and conditions of ToL material reuse and
redistribution, please see the Tree of Life Copyright
Policies.
Page: Tree of Life
Turbinoliidae .
Authored by
Stephen D. Cairns.
The TEXT of this page is licensed under the
Creative Commons Attribution-NonCommercial License - Version 3.0. Note that images and other media
featured on this page are each governed by their own license, and they may or may not be available
for reuse. Click on an image or a media link to access the media data window, which provides the
relevant licensing information. For the general terms and conditions of ToL material reuse and
redistribution, please see the Tree of Life Copyright
Policies.
- First online 28 October 2002
Citing this page:
Cairns, Stephen D. 2002. Turbinoliidae . Version 28 October 2002. http://tolweb.org/Turbinoliidae/19024/2002.10.28 in The Tree of Life Web Project, http://tolweb.org/
 Go to quick links
Go to quick search
Go to navigation for this section of the ToL site
Go to detailed links for the ToL site
go to top
top
Go to quick links
Go to quick search
Go to navigation for this section of the ToL site
Go to detailed links for the ToL site
go to top
top