Polycystina
Polycystine radiolarians
Jane K. Dolven, Kjell Rasmus Bjørklund, Jon Bråte, Anders K. Krabberød, Dag Klaveness, and Kamran Shalchian-Tabrizi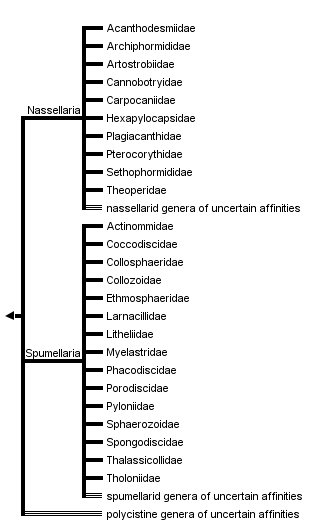

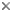
This tree diagram shows the relationships between several groups of organisms.
The root of the current tree connects the organisms featured in this tree to their containing group and the rest of the Tree of Life. The basal branching point in the tree represents the ancestor of the other groups in the tree. This ancestor diversified over time into several descendent subgroups, which are represented as internal nodes and terminal taxa to the right.

You can click on the root to travel down the Tree of Life all the way to the root of all Life, and you can click on the names of descendent subgroups to travel up the Tree of Life all the way to individual species.
For more information on ToL tree formatting, please see Interpreting the Tree or Classification. To learn more about phylogenetic trees, please visit our Phylogenetic Biology pages.
close boxIntroduction
Polycystine radiolarians have enchanted scientists with their astonishing beauty ever since they were first discovered in the marine plankton in the nineteenth century. Their tiny skeletons – with often fascinating intricate shapes – have been interesting not only to natural scientists, but also to architects (like A. Gaudi and R. Binet), painters, jewelers, quilters, sculpturers and musicians who have used them as inspiration for their work. With fossil representatives extending continuously back to the Cambrian, i.e. more than 500 million years, few other microorganisms have existed longer and left behind a more detailed evolutionary record than radiolarians.
Many fossil radiolarian species have robust skeletons that preserve well in the sediments and distinct morphologies that are easily recognized under the microscope. Some of these also have a rapid evolutionary record and a widespread distribution making them excellent index fossils (biostratigraphic markers). They are therefore extensively used to date and correlate sediments and rocks in the Integrated Ocean Drilling Program and the petroleum industry. Fossil radiolarian assemblages are frequently used to reconstruct ocean temperatures throughout the Quaternary (Pisias et al. 1997; Dolven et al. 2002). These paleoclimatic reconstructions are based on the assumption that the living assemblages are modern analogs to the fossil ones, and that their current correlation with temperature or other environmental parameters has been consistent through time. Moreover, radiolarians have been used to prove plate tectonics and the formation of the Himalayas due to their presence in uplifted marine rocks now located at more than 5500 m above sea level.
So, who are these interesting and useful small creatures?
Radiolarians are marine planktonic unicellular eukaryotes. They are present in all oceans at all depths. Most species occur solitarily, ranging in size between 30 and 300 µm, although some colonial species are also known. The latter consist of numerous individuals enclosed in a gelatinous matrix and may reach up to a couple of meters in length (Anderson 1983).
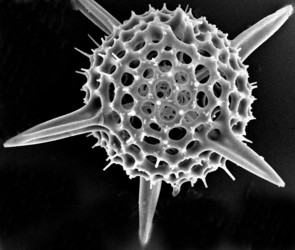
Fig. 1. The test of Hexacontium pachydermum Jørgensen 1900. © Jane K. Dolven
Although it is the possession of a central capsulea that traditionally has been used to distinguish radiolarians from other protists, they are perhaps more commonly known for their siliceous skeleton (test). This test, which is present in most species, can be found in a large variety of shapes, from simple spicules to extremely intricate architectural features – more complex than what is found in any other protist organism.
Based on similarities and differences in skeletal elements and structures (e.g. shell geometry, number of segments or concentric shells, presence and number of spines, etc.) several thousand species (ca. 8000 according to De Wever et al. 2001) have been described, including both fossil and living forms, and new species are discovered every year. Of these, about 500 morphospecies live and thrive in the oceans today. However, the DNA sequencing of marine samples indicates a larger diversity (Not et al. 2007).
Radiolarians have been an important marine plankton group since the early Phanerozoic (about 500 million years before present), and are still a significant component of the pelagic environment today. As with most planktonic organisms, their diversity and abundance in a geographical region is related to variables such as temperature, salinity, productivity, and availability of nutrients. Highest abundance and diversity of radiolarians is found in the tropical and subtropical regions, especially in water masses rich in nutrients (e.g. upwelling areas). They are also usually more abundant in the upper 100 m than below, possibly due to their frequent association with symbiotic algae.
a The presence of a central capsule has traditionally been used as one of the main criteria for placing the polycystines, acantharians and phaeodarians together in the group of Radiolaria (Haeckel 1887). Acantharia was later removed from the group due to differences in chemical composition and skeletal morphology (Deflandre 1952). However, recent DNA analysis suggests a close relationship between acantharians and solitary spumellarians (Polet et al, 2004; Yuasa et al. 2006), suggesting that acantharians perhaps again should be placed within Radiolaria. Molecular analysis of phaeodarians, on the other hand, shows that these are cercozoans and do not belong with the radiolarians.
Characteristics
Cellular organization
Polycystine radiolarians are characterized by having a central capsule that divides the protoplasm into an endoplasm and ectoplasm (Anderson 1983). The central capsule contains one or more nuclei, large mitochondria, the Golgi apparatus, lysosomes, endoplasmatic reticulum, and ribosomes, as well as vacuoles, lipid droplets and food reserves (Anderson 1983; Cachon et al. 1990). It also contains a specialized microtubule organizing center, called an axoplast from which the axopodia (slender, ray-like cytoplasmic extensions stiffened by rigid axial filaments) arise. Functions like reproduction, respiration, and biochemical synthesis are all carried out inside the central capsule. The shape of the central capsule varies from species to species.
The extracapsular body contains the ectoplasm, including digestive vacuoles, alveoli and symbionts if present, and the pseudopodia which are cytoplasmic projections outside the cell (Anderson 1983). The latter comprises of rhizopodia (fine, very delicate branching or reticulated pseudopodia), radial axopodia, and delicate filopodia. Some spumellarian species possess an axoflagellum, i.e. a projection formed by several lateral fused axopodia passing through a specialized pore in the central capsular wall. Nassellarians do not have an axoflagellum, but instead an axial projection and several other characteristic pseudopodial extensions (Sugiyama et al. 2008).
The central capsular membrane is chitinous or pseudochitinous (De Wever et al. 2001). It is also pigmented and therefore relatively easy to recognize in living specimens. Pores (fusules) and capsular fissures passes through the membrane connecting the endoplasm with the extoplasm. It is also through the fusules that the axopodia extends. The distribution patterns of fusules on the capsular membrane are often used to differentiate spumellarians from nassellarians, as the former have numerous evenly distributed pores while the latter have pores concentrated on only one part.
The radiolarian skeleton
Radiolarians are characterized by their siliceous (SiO2 x nH2O) skeleton, absent only in a few species. When alive, the skeleton (test) is completely embedded in the cell’s cytoplasm (i.e. inside the cell membrane) to protect it from dissolution in the seawater. The ectoplasm stores most of the skeleton, though there are some examples (of spumellarians and entactinarians) where the innermost test parts are located in the endoplasm. The radiolarian skeleton is solid and can therefore be distinguished from siliceous sponge spicules which skeletons have a central tube.
The two most important types of elements that make up the radiolarian skeleton are the bars (elongated elements connected at both ends) and spines (elongated elements attached only at one end) (figs. 2-4; Kling 1978). The variation of skeletal shapes among radiolarians is remarkable, with each species having their own characteristics. Some have only very simple elements (spicules) while others display amazingly complex constructions, especially when taking into account their small size. Characters often used to divide radiolarians into groups are: symmetry, presence or absence of spheres, number of segments, internal structures, pores, spines and wall type. Of the latter three types are known: 1) Latticed wall, 2) Spongy wall, and 3) Perforate plate wall.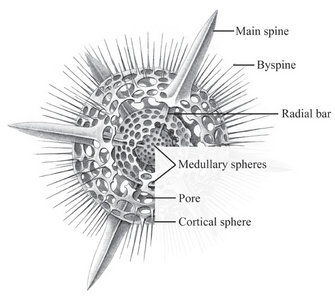 Fig. 2. Spumellaria. Modified from Haeckel 1862 (Pl. 23, fig. 6). © Jane K. Dolven | 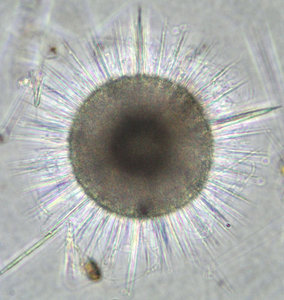 Fig. 3. Living Hexacontium sp. © Jane K. Dolven |
Spumellarians and entactinarians are recognized by their radial symmetry, and for often being spherical and having radiating spines (figs. 2-3). If there is more than one sphere present, as is often the case in spumellarian skeletons, the outermost (external) one is called the cortical sphere and the inner ones medullary spheres. The spheres are connected by radial bars that may vary in thickness. These bars always start on the surface of the innermost medullary sphere extending outwards, and never meet in the center of the specimen (which is the case for the acantharians). Two types of spines are common; 1) the main spines which are “external projections” of the radial bars outside the cortical sphere, and 2) the byspines which are spines that starts on the surface of the cortical sphere without being supported by inner radial bars. Other common spumellarian shapes in addition to the spherical are the discoidal, ellipsoidal and spiral forms.
The symmetry of nassellarians is usually bilateral. Common to all nassellarians is a spicule (fig. 4), consisting of a median bar with a number of extending spines. The most important are the apical (A) and dorsal (D) spines on one end of the bar and the ventral (V) and lateral (L) spines on the other end of the bar. Sometimes the spines are connected by arches, as in the sagittal ring where the ventral and apical spine is joined. The simplest form of nassellarians skeletons consist only of a spicule. In more complex and developed forms an outer skeleton is formed around the spicule, consisting of one or more segments (fig. 5). The first segment, which also contains the internal spicule, is called the cephalis. This segment may have various shapes (spherical or elongate), lobes formed by the internal spicule arches, pores, and do sometimes possess horns, tubes and/or additional spines. The second segment is referred to as the thorax. When spines are projecting from this segment they are called wings. The third segment is the abdomen and all other following segments are called post-abdominal. Like the cephalis, the thorax and abdomen often have pores with various shapes, sizes, and arrangements, and sometimes characteristic furrows or striations. The terminal segment may be closed or have an aperture. It can sometimes also have a basal ring called the peristome (with or without small teeth), and terminal projections referred to as feet.
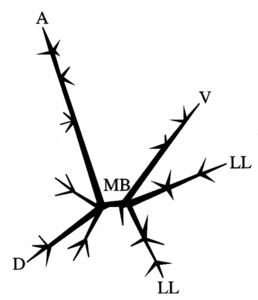 Fig. 4. Nassellaria spicule. Modified from De Wever et al. (2001). © Jane K. Dolven | 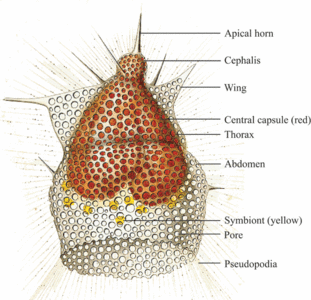 Fig. 5. Nassellaria. Modified from Haeckel 1862 (Pl. 8, fig. 1). © Jane K. Dolven |
The benefits of having a skeleton are many. The skeleton may serve as protection for the radiolarian cell and its symbionts when present. It can also serve as ballast in the water masses as opposed to the frothy, bubble-like alveoli which keep the cell floating. Radially arranged spines may also provide anchorage, support and stability for radiating rhizopodia increasing the rhizopodial grip when the radiolarian is capturing prey. However, why some radiolarians are putting so much energy into building these incredible complex siliceous constructions are yet to be discovered.
Reproduction
Current knowledge on radiolarian reproduction is very limited and so far restricted only to a very few of the hundreds of species living in the oceans today. One of the reasons for this is the problems associated with keeping radiolarians alive in the laboratory, and in particular getting them to reproduce. In the literature both asexual and sexual reproductions have been proposed (summarized in Anderson 1983). And even dimorphic, sexual-asexual, alternating generations analogous to the reproduction stages of foraminifera has been suggested (Kling 1971). The latter is based on interesting plankton data, but is not confirmed by any laboratory observations.
Reproduction processes observed so far are: 1) binary fission, found in colonial spumellarians, in particular those who lack skeletons or possess only spicules; and 2) multiple fission with the production of tiny (smaller than 10 µm) swarmer cells, found in both solitary and colonial Spumellaria (Anderson 1983). It has not yet been firmly established whether these swarmers are gamets or agamets.
As regarding the swarmer production two phenomena are of particular interest:
- The swarmers are small, pointed flagellated cells with two undulipodia (Cachon et al. 1990) and a tiny crystal inside (Anderson 1983). The intriguing thing about this crystal is that it seems to be rich in strontium (Anderson 1983), which is also one of the major chemical components of acantharians skeleton (SrSO4). Acantharians were previously considered to be radiolarians (Haeckel 1887) due to their common perforated central capsule, but were later removed because of their differences in chemical composition and skeletal morphology. The presence of strontium in the radiolarian swarmers suggests an evolutionary relationship with acantharians. The close relationships between polycystine radiolarians and acantharians has also recently been documented by molecular (18S rDNA) studies (López-García et al. 2002; Yuasa et al. 2006).
- Prior to the release of swarmers, the mother cell (or “mother colony”) looses buoyancy (by e.g. shedding the alveolar extracapsulum or increasing density by contraction of the colony) and starts to sink. How deep the mother cell(s) descends before the swarmers are set free is unknown, but for some radiolarian species a water depth of 400-600 m have been proposed (Anderson 1983). Recently, high percentages (~20%) of radiolarian DNA have been recorded in some deep water (500-3000 m) environmental samples (Not et al. 2007). These environmental samples are based on the analysis of size-fractionated samples <2 µm, and therefore likely to exclude the normal sized radiolarian (30-300 µm). Radiolarian swarmers, on the other hand, could due to their small size be included and may explain the high percentage of radiolarian DNA in these deepwater samples.
As far as we know, no culturing projects have ever succeeded in transforming swarmers into small radiolarian offspring, so the swarmers’ ultimate role in the reproductive cycle is still unknown. Due to the limited data, it is neither known which of the described reproductions processes (or possibly others not yet described) that are the most common for radiolarians, nor what factors that are initiating the reproduction. More research on the topic is therefore needed to shed new light on their evolutionary history. The possibility of multiplying radiolarians in the laboratory may also play a vital role in future molecular investigations as the “single-specimen-analysis” so far used have proven difficult.
Longevity
Since it is not possible to observe a full radiolarian life cycle in the laboratory it is difficult to give accurate information on their life span. However, there are two ways that are commonly used to estimate it: 1) by recording maximum survival time of radiolarian species in the laboratory; and 2) by comparing number of specimens in the plankton with accumulation rates of skeletons in the sediments (e.g. sediment traps). The longest survival time recorded in the laboratory is about 60 days (pers comm. Atsushi Matsuoka, InterRad XII 2009). Regarded that the specimen was already a juvenile at the time of capture in the plankton the longevity of this specimen can be extrapolated even further. Using the second method, Casey et al. (1971) found a turnover time of about a month. Based on these two estimates it seems reasonable to assume that radiolarians live between 1-2 months, but this may of course vary from species to species and also depend on the season and factors such as temperature, salinity and food availability.
Prey and feeding strategies
Radiolarians capture and digest a large variety of small prays (e.g. silicoflagellates, ciliates, tintinnids, diatoms, crustacean larvae, copepods and bacteria). This is done by use of their “sticky” pseudopodia (in particular the axopodia and/or rhizopodia; figs. 6-7). Several different feeding strategies have been reported for radiolarians (Anderson 1983; Matsuoka 2007). The captured food is enclosed in a digestive vacuole and carried into the ectoplasm where the digestion occurs. Although many radiolarian species are assumed to be omnivores, some feeding preferences do exist. There are, for instance, species preferring algal- before animal prey and vise versa (Anderson 1983). Some radiolarians are not actively hunting their food, but live primarily of nutrients obtained from their symbiotic photosynthesizing algae.
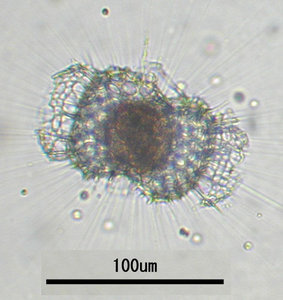 Fig. 6. Living Didymocyrtis tetrathalamus (Haeckel 1887) with pseudopodia. © Tomoko Yuasa | 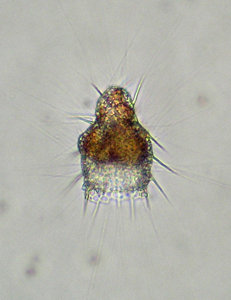 Fig. 7. Living Lithomelissa setosa Jørgensen 1900 with pseudopodia. © Jane K. Dolven |  Fig. 8. Living Euchitonia elegans (Haeckel 1887) with symbionts (yellow). © Tomoko Yuasa |
Symbiosis (and Parasitism)
Polycystine radiolarians are often living in association with other microorganisms (fig. 8). The most commonly found are the dinoflagellates, but prymnesiophyte and prasinophyte associations have also been observed, though not at the same time (Anderson 1983). When hosting symbiotic algae, these are mainly kept within the polycystine ectoplasm and held within the rhizopodial network surrounding the central cell body. Laboratory studies have shown that these symbionts are often distributed in the outer cell periphery during daylight and repositioned around the capsular wall during darkness (Anderson 1983). Experiments have also shown that radiolarians containing phototrophic algae survive longer in nutrient-sparse water than those without, suggesting that the symbionts play a nutritive role in the radiolarian life (Anderson 1983). The radiolarian, on the other hand, may provide to the symbiont protection, nutrients (mostly waste material containing nitrogen and phosphorus) and carbon dioxide required for photosynthesis.
The number and density of symbionts seem to be dependent upon radiolarian species and their size. Some large solitary and colonial forms can possess up to several thousands at the same time (Anderson 1983). The increasing number of symbionts with radiolarian growth, taken together with the observations of various states of protist divisions within the radiolarian cytoplasm, indicates that these associates reproduce and grow within the radiolarian host (Anderson 1976; De Wever et al. 2001). This relationship ends immediately prior to radiolarian reproduction or death with a digestion or expulsion of the algae.
Some radiolarian-associates are of a more parasitic nature. The “nastiest” ones are the heterotrophic dinoflagellate species, Merodinium and Solenodinium (Anderson 1983), and possibly also Amoebophrya (Dolven et al. 2007) whose invasions have a lethal outcome for the radiolarian.
Mobility
Radiolarians are largely non-motile, pelagic organisms, well adapted to a planktonic mode of life. They have intracapsular vacuoles filled with lipids or other buoyancy-enhancing fluids that possibly serve as floating mechanisms, and a “heavy” siliceous skeleton that functions as ballast. As they only have flagella when they are swarmers, their main way of moving from one place to another as adults is being passive passenger of ocean currents. It is, however, likely that radiolarians can move vertically in the water masses by regulating (either by expansion or contractions) their extracapsular alveolar volume. E.g. Brandt (1895) suggested that these frothy, bubble-like alveoli contain CO2-saturated seawater and that the radiolarian could control their buoyancy by adjusting the CO2-content. Pokorný (1963) took this idea one step further and suggested that the regulation of CO2 is related to photosynthetic activity of radiolarian symbionts causing a diurnal vertical migration. Even though the hypothesis of vertical movement has not yet been proved, it is supported by the fact that some radiolarian species shed their extracapsulum to loose buoyancy and sink in the early phase of reproduction or when they are somehow threatened and need to escape.
References
Anderson, O.R., 1976. A cytoplasmic fine-structure study of two spumellarian Radiolaria and their symbionts. Marine Micropaleontology, 1: 81-99.
Anderson, O.R., 1983. Radiolaria. Springer-Verlag, New York, 355 pp.
Brandt, K., 1895. Biologische und faunistische Untersuchungen an Radiolarien und anderen pelagischen Thieren. I. Untersuchungen uber den hydrostatischen Apparat von Thalassicollen und Koloniebildenden Radiolarien. Zoologische Jahrbucher, Systematik, Okologie und Geographie der Thiere, 9: 27-74.
Cachon, J., Cachon, M. and Estep, K.W., 1990. Phylum Actinopoda Classes Polycystina [= Radiolaria] and Phaeodaria. Handbook of Protoctista, Margulis, L., Corliss, J. O., Melkonian, M. and Chapman, D. J. [eds.], Jones and Bartlett [The Jones and Bartlett Series in Life Sciences], Boston, USA, 334-346.
Casey, R.E., Partridge, T.M. and Sloan, J.R., 1971. Radiolarian life spans, mortality rates and seasonality gained from sediment and plankton samples. Proceedings of the II Planktonic Conference, Roma 1970, vol. , Farinacci, A. [ed.], Edizioni Tecnoscienza, Roma, Italy, 159-165.
Deflandre, G., 1952. Classe des Radiolaires. Traite de Paleontologie, vol. 1, Piveteau, J. [ed.], Masson, Paris, France, 303-313.
De Wever, P., Dumitrica, P., Caulet, J.P., Nigrini, C. and Caridroit, M., 2001. Radiolarians in the sedimentary record. Gordon and Breach Science Publishers, Amsterdam, The Netherlands, 533 pp.
Dolven, J. K., Cortese, G. and Bjørklund, K. R., 2002. A high-resolution radiolarian derived paleotemperature record for the Late Pleistocene-Holocene in the Norwegian Sea. Paleooceanography, 17(4): 1072 (13 pages).
Dolven, J.K., Lindqvist, C., Albert, V.A., Bjørklund, K.R., Yuasa, T., Takahashi, O. and Mayama, S., 2007. Molecular diversity of alveolates associated with neritic North Atlantic radiolarians, Protist, 158: 65-76.
Haeckel, E., 1887. Report on the Radiolaria collected by the H.M.S. Challenger during the Years 1873-1876. Report on the Scientific Results of the Voyage of the H.M.S. Challenger, Zoology 18: 1803 pp.
Kling, S. A., 1971. Dimorphism in Radiolaria. Proceedings of the II Planktonic Conference, Roma 1970, vol. 1, Farinacci, A. [ed.], Edizioni Tecnoscienza, Roma, Italy, 663-667.
Kling, S. A., 1978. Radiolaria. Introduction to Marine Micropaleontology, Haq, B. U. and Boersma, A. [eds.], Elsevier Scientific Publishing Company, New York, USA, 203-244.
López-García, P., Rodríguez-Valera, F., Pedrós-Alió, C. and Moreira, D., 2001. Unexpected diversity of small eukaryotes in deep-sea Antarctic plankton. Nature 409: 603-607.
Matsuoka, A., 2007. Living radiolarian feeding mechanisms: new light on past marine ecosystems. Swiss j. geosci. 100: 273279.
Not, F., Gausling., R., Azam, F., Heidelberg, J.F., Worden, A.Z, 2007. Vertical distribution of picoeukaryotic diversity in the open ocean. Environmental Microbiology 9:12331252.
Pokorný, V., 1963. Principles of Zoological Micropaleontology. Pergamon Press, London, 652 pp.
Polet, S., Berney, C., Fahrni, J, and Pawlowski, J, 2004. Small subunit ribosomal RNA gene sequences of Phaeodarea challenge the monophyly of Haeckels Radiolaria. Protist, 155: 53-63.
Pisias, N.G., A. Roelofs, and Weber, M., 1997. Radiolarian-based transfer functions for estimating mean surface ocean temperatures and seasonal range. Paleoceanography, 12: 365379.
Sugiyama, K., Hori, R.S., Kusunoki, Y. and Matsuoka, A, 2008. Pseudopodial features and feeding behavior of living nassellarians Eucyrtidium hexagonatum Haeckel, Pterocorys zancleus (Müller) and Dictyocodon prometheus Haeckel. Paleontological Research, 12(3): 209222.
Yuasa, T., Takahashi, O., Dolven J.K., Mayama, S., Matsuoka, A., Honda, D-, Bjørklund K.R., 2006. Phylogenetic position of the small solitary phaeodarians (Radiolaria) based on 18S rDNA sequences by single cell PCR analysis. Marine Micropaleontology, 59: 104-114.
Title Illustrations

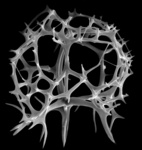
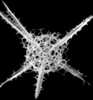
| Scientific Name | Rhizoplegma boreale (Cleve 1899) |
|---|---|
| Specimen Condition | Fossil -- Period: Holocene |
| Identified By | JKD |
| Copyright |
© Kjell Rasmus Bjørklund

|
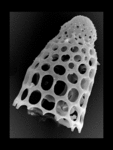
| Scientific Name | Lamprocyclas maritalis Haeckel 1887 |
|---|---|
| Specimen Condition | Fossil -- Period: Holocene |
| Identified By | JKD |
| Copyright |
© Kjell Rasmus Bjørklund

|
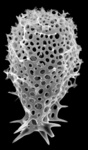
| Scientific Name | Euchitonia elegans Haeckel, 1887 |
|---|---|
| Specimen Condition | Fossil -- Period: Holocene |
| Identified By | KRB |
| Copyright |
© Kjell Rasmus Bjørklund

|
| Scientific Name | Nephrospyris knutheieri Goll and Bjørklund 1985 |
|---|---|
| Specimen Condition | Fossil -- Period: Holocene |
| Identified By | KRB |
| Copyright |
© Kjell Rasmus Bjørklund

|
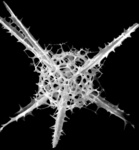
| Scientific Name | Artostrobus joergenseni Petrushevskaya 1967 |
|---|---|
| Specimen Condition | Fossil -- Period: Holocene |
| Identified By | JKD |
| Image Use |
 This media file is licensed under the Creative Commons Attribution-NonCommercial-ShareAlike License - Version 3.0. This media file is licensed under the Creative Commons Attribution-NonCommercial-ShareAlike License - Version 3.0.
|
| Copyright |
© Jane K. Dolven

|
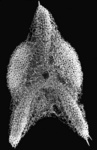
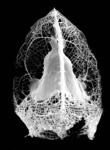
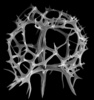
| Scientific Name | Callimitra carolotae |
|---|---|
| Specimen Condition | Fossil |
| Identified By | KRB |
| Copyright |
© Kjell Rasmus Bjørklund

|
| Scientific Name | Mitrocalpis araneafera Popofsky 1908 |
|---|---|
| Specimen Condition | Fossil |
| Identified By | KRB |
| Copyright |
© Kjell Rasmus Bjørklund

|
About This Page
This page is being developed as part of the Tree of Life Web Project Protist Diversity Workshop, co-sponsored by the Canadian Institute for Advanced Research (CIFAR) program in Integrated Microbial Biodiversity and the Tula Foundation.
Jane K. Dolven

Natural History Museum, University of Oslo, Norway
Kjell Rasmus Bjørklund

Naturhistorisk Museum, Universitetet i Oslo, Norway
Jon Bråte

University of Oslo, Norway
Dag Klaveness

Universitetet i Oslo, Norway
Kamran Shalchian-Tabrizi

University of Oslo, Oslo, Norway
Correspondence regarding this page should be directed to Jane K. Dolven at , Kjell Rasmus Bjørklund at , Jon Bråte at , Anders K. Krabberød at , Dag Klaveness at , and Kamran Shalchian-Tabrizi at
Page copyright © 2009 Jane K. Dolven, Kjell Rasmus Bjørklund, Jon Bråte, , Dag Klaveness, and Kamran Shalchian-Tabrizi
 Page: Tree of Life
Polycystina. Polycystine radiolarians.
Authored by
Jane K. Dolven, Kjell Rasmus Bjørklund, Jon Bråte, Anders K. Krabberød, Dag Klaveness, and Kamran Shalchian-Tabrizi.
The TEXT of this page is licensed under the
Creative Commons Attribution-NonCommercial License - Version 3.0. Note that images and other media
featured on this page are each governed by their own license, and they may or may not be available
for reuse. Click on an image or a media link to access the media data window, which provides the
relevant licensing information. For the general terms and conditions of ToL material reuse and
redistribution, please see the Tree of Life Copyright
Policies.
Page: Tree of Life
Polycystina. Polycystine radiolarians.
Authored by
Jane K. Dolven, Kjell Rasmus Bjørklund, Jon Bråte, Anders K. Krabberød, Dag Klaveness, and Kamran Shalchian-Tabrizi.
The TEXT of this page is licensed under the
Creative Commons Attribution-NonCommercial License - Version 3.0. Note that images and other media
featured on this page are each governed by their own license, and they may or may not be available
for reuse. Click on an image or a media link to access the media data window, which provides the
relevant licensing information. For the general terms and conditions of ToL material reuse and
redistribution, please see the Tree of Life Copyright
Policies.
- First online 20 October 2009
- Content changed 29 November 2009
Citing this page:
Dolven, Jane K., Kjell Rasmus Bjørklund, Jon Bråte, Anders K. Krabberød, Dag Klaveness, and Kamran Shalchian-Tabrizi. 2009. Polycystina. Polycystine radiolarians. Version 29 October 2009 (under construction). http://tolweb.org/Polycystine_radiolarians/121189/2009.10.29 in The Tree of Life Web Project, http://tolweb.org/










 Go to quick links
Go to quick search
Go to navigation for this section of the ToL site
Go to detailed links for the ToL site
Go to quick links
Go to quick search
Go to navigation for this section of the ToL site
Go to detailed links for the ToL site